Vial Packaging: Definition, Process, and Its Role in the Pharmaceutical Industry
Vial packaging plays a crucial role in maintaining the safety, stability, and effectiveness of injectable medicines throughout their lifecycle. Because healthcare professionals administer injectable drugs directly into the body, manufacturers must keep them sterile and free from contamination until use. Even small changes in the packaging environment can compromise drug quality, so pharmaceutical manufacturers design packaging systems that follow strict safety, quality, and regulatory standards.
The pharmaceutical industry plays a crucial role in global healthcare by developing and delivering medicines that treat diseases, prevent infections, and improve quality of life. Among the many forms of medicines available today. Pharmaceutical injectables are one of the most important and widely used dosage forms. Healthcare professionals administer these medications directly into the human body using syringes, infusion systems, or injection devices. Because injectable medicines bypass the digestive system and enter the bloodstream or tissues directly, they act quickly and provide high therapeutic effectiveness.
However, because injectable drugs are delivered directly into the body, they require extremely strict manufacturing, storage, and packaging conditions. Even minor contamination or degradation can affect drug safety and effectiveness. For this reason, pharmaceutical manufacturers rely on specialized containers and advanced packaging technologies to maintain the stability, sterility, and quality of injectable medications. Among the various packaging options available, vials are one of the most widely used containers in the pharmaceutical industry.
What Are Pharmaceutical Injectables?
Pharmaceutical injectables are sterile medications that healthcare professionals administer directly into the body through injections or intravenous infusions. These drugs are commonly used in hospitals, clinics, and emergency care settings because they provide rapid therapeutic action. Unlike oral medications, injectable drugs bypass the digestive system and enter the bloodstream directly, allowing them to act faster and deliver precise dosages.
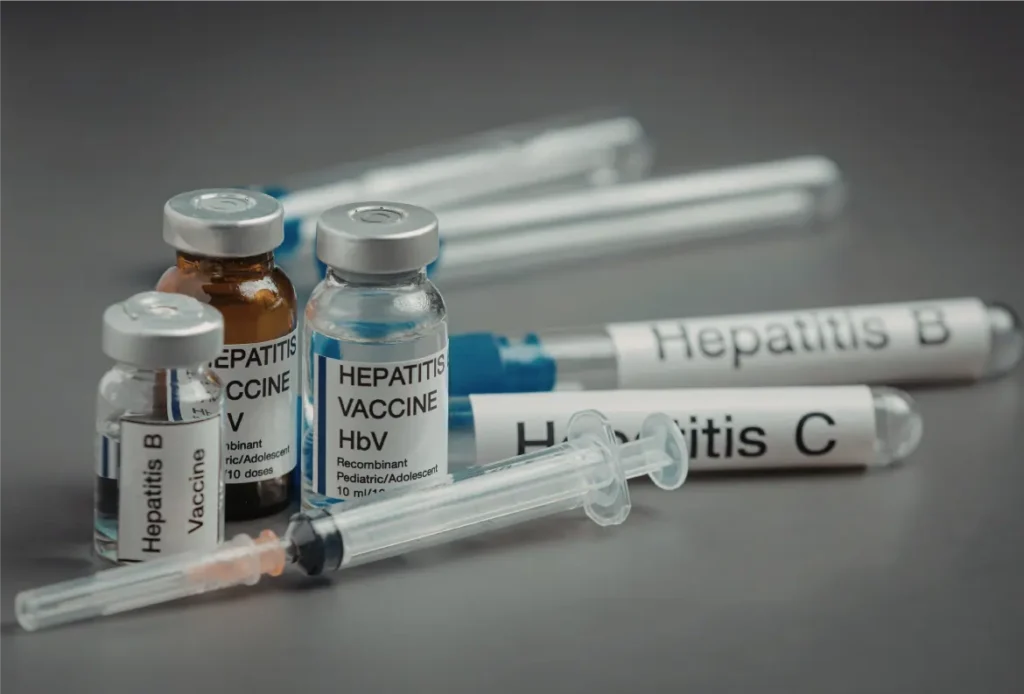
Injectable medicines are widely used for a variety of treatments. For example, vaccines, insulin, antibiotics, biologics, and emergency medications are often delivered through injections. These drugs require highly controlled manufacturing environments because sterility is essential to ensure patient safety.
Furthermore, injectable drugs may be formulated as liquids, suspensions, or powders that require reconstitution before administration. Because of this variety in formulations, pharmaceutical manufacturers must carefully select appropriate containers that protect the drug while maintaining its stability throughout its shelf life.
What Are Vials in the Pharmaceutical Industry?
A vial is a small container or bottle used to store pharmaceutical products in liquid, powder, or lyophilized form. Traditionally, vials were made from glass, but modern pharmaceutical containers may also be manufactured from high-quality plastics such as polypropylene. These containers are designed to maintain sterility and protect medicines from environmental factors such as light, moisture, air, and contamination.
Vial-like glass containers have existed since classical antiquity, demonstrating their long-standing role in medicine and laboratory practices. Today, pharmaceutical vials remain essential for storing injectable drugs, vaccines, diagnostic reagents, and laboratory samples.

Vials are available in different designs depending on their intended use. Some are single-dose vials, which contain a single quantity of medication that must be used immediately after opening. Others are multi-dose vials, which allow healthcare professionals to withdraw multiple doses from the same container. Multi-dose vials typically include preservatives to maintain sterility after repeated use.
Pharmaceutical Vial Packaging: Types, Applications, and Packaging Considerations
One of the primary functions of pharmaceutical vial packaging is to protect injectable medicines during storage, transportation, and handling. Pharmaceutical products often travel long distances from manufacturing facilities to hospitals, clinics, pharmacies, and laboratories around the world. During this journey, the product may be exposed to various environmental conditions such as temperature fluctuations, humidity, light exposure, and mechanical vibrations. Well-designed pharmaceutical vial packaging protects the container from these conditions and helps ensure that the drug remains stable and effective until it reaches healthcare providers and patients.
Pharmaceutical manufacturers also design packaging systems to protect products from mechanical damage during transportation and distribution. Vials, particularly glass containers, can be fragile and vulnerable to breakage if they are not properly supported. To address this challenge, pharmaceutical packaging systems often include secondary protective materials such as trays, cartons, inserts, and cushioning components. These elements stabilize the vials during transportation and reduce the risk of damage caused by movement, impact, or vibration. As a result, strong vial packaging solutions are essential for ensuring the safe delivery of injectable drugs.
Packaging Considerations
In addition to protecting the drug from environmental factors, vial packaging must also maintain sterility and container closure integrity. Injectable medications require sterile packaging because contamination could lead to serious health risks for patients. The packaging system must create a secure barrier that prevents microorganisms, dust, or other contaminants from entering the vial. This is especially important for injectable drugs that may remain in storage for long periods before use. A reliable pharmaceutical vial packaging system, therefore, ensures that sterility is maintained from the manufacturing stage to the point of administration.
Another important advantage of proper packaging is improved efficiency during storage and distribution. Well-organized packaging systems allow healthcare professionals and distributors to handle and store vials safely and efficiently. Structured packaging formats make it easier to identify products, monitor batch numbers, and manage inventory. Clear labelling and organized packaging also help maintain regulatory compliance and improve traceability throughout the pharmaceutical supply chain.
Furthermore, packaging design plays a significant role in supporting automated pharmaceutical manufacturing processes. Modern pharmaceutical production facilities rely heavily on automated systems for filling, sealing, labelling, and inspecting vials. Proper packaging formats allow vials to move smoothly through these automated systems without disruption. This improves production efficiency, reduces human intervention, and helps maintain consistent quality standards.
Importance of Vials in Drug Stability and Safety
Vials are not simply storage containers; they are critical components of pharmaceutical drug delivery systems. The selection of the right vial is essential for maintaining the stability and integrity of the medication throughout its lifecycle. A poorly designed container can lead to contamination, chemical reactions, or degradation of the drug.
Pharmaceutical vials protect medications from environmental conditions such as light exposure, oxygen, humidity, and temperature fluctuations. At the same time, they must maintain sterility and prevent microbial contamination. In addition, vial materials must be chemically compatible with the drug formulation to avoid interactions that could compromise drug quality.
Therefore, pharmaceutical manufacturers carefully evaluate container materials, manufacturing methods, and closure systems before selecting a vial for specific applications.
Types of Vials Used in the Pharmaceutical Industry

Manufacturers categorize pharmaceutical vials based on manufacturing method, material composition, size, colour, closure type, and base design. Each type provides unique advantages depending on the drug formulation and storage requirements.
Vials Based on Manufacturing Method
Manufacturers produce pharmaceutical vials using two primary manufacturing methods: moulded vials and tubular vials. Manufacturers create moulded vials by melting glass and shaping it inside molds. This process produces containers with thicker walls and strong mechanical stability. Because of their durability, manufacturers use moulded vials for products that require robust packaging and protection during transportation.
In contrast, manufacturers produce tubular vials by heating and shaping glass tubing into containers. This method creates vials with uniform wall thickness and high dimensional accuracy. Manufacturers commonly use tubular vials for high-precision pharmaceutical products, including diagnostic reagents and sensitive biomedical formulations.
Vials Based on Material
Manufacturers typically produce vials using either glass or plastic materials. Each material provides specific advantages depending on the application. Manufacturers widely prefer glass vials for injectable drugs because glass remains chemically inert and resists temperature changes. Borosilicate glass, in particular, resists chemical reactions and thermal stress, which makes it ideal for pharmaceutical use.
Manufacturers increasingly use plastic vials in certain applications because they offer a lightweight design and strong resistance to breakage. Materials such as polypropylene and polyethylene provide good chemical resistance and support laboratory and diagnostic applications.
The choice between glass and plastic often depends on factors such as cost, durability, environmental considerations, and compatibility with the drug formulation.
Vials Based on Size
Pharmaceutical vials are available in a wide range of sizes to accommodate different dosage requirements, drug formulations, and clinical applications. The size of a vial plays an important role in ensuring accurate dosing, maintaining drug stability, and improving efficiency during storage, transportation, and packaging. Manufacturers determine vial sizes based on factors such as the required dose, filling volume, method of administration, and whether the medication is intended for single-dose or multi-dose use. Selecting the appropriate vial size also helps reduce drug wastage and supports the smooth operation of automated filling and packaging systems used in pharmaceutical manufacturing.
Manufacturers typically produce small vials ranging from 1 mL to 5 mL and use them for vaccines, high-potency drugs, and specialty injectable formulations that require precise dosing. They produce medium-sized vials ranging from 5 mL to 20 mL and widely use them for injectable antibiotics, diagnostic reagents, and lyophilized drugs that require reconstitution before administration. Larger vials, generally ranging from 25 mL to 100 mL, are suitable for medications that require higher fill volumes or multiple doses, such as hospital treatments and intravenous solutions. In addition to volume capacity, manufacturers must also consider factors such as headspace requirements, compatibility with filling equipment, and storage efficiency to ensure optimal performance throughout the pharmaceutical packaging process.
Vials Based on Colour
The colour of pharmaceutical vials plays an important role in protecting drug formulations from environmental factors, particularly light exposure. Many pharmaceutical compounds are sensitive to ultraviolet (UV) rays and visible light, which can cause chemical reactions that reduce the drug’s stability and effectiveness. Therefore, selecting the correct vial colour is an essential consideration during pharmaceutical packaging design. Manufacturers commonly use clear vials for formulations that are not sensitive to light because they allow easy visual inspection of the contents. This transparency helps healthcare professionals and quality control teams identify issues such as discoloration, particles, or precipitation before use. As a result, manufacturers widely use clear vials for injectable solutions, laboratory reagents, and diagnostic products.
On the other hand, amber vials are designed to protect pharmaceutical products from harmful light exposure. Amber glass contains pigments that absorb ultraviolet and blue light, preventing these wavelengths from reaching the drug inside the container. This protection helps reduce the risk of photodegradation, which can occur when light alters the chemical structure of a drug. Amber vials are therefore widely used for photosensitive medicines such as certain antibiotics, vitamins, and biologics. By shielding the product from light, amber containers help maintain drug stability, extend shelf life, and ensure the medication remains safe and effective until administration.
Vials Based on Closure and Finish
The design of the vial opening, commonly known as the finish, determines how manufacturers seal the container and how effectively it protects the pharmaceutical product inside. The finish allows the vial closure system to form a secure seal that maintains sterility, prevents contamination, and protects the drug from environmental exposure. In pharmaceutical applications, manufacturers must maintain container closure integrity to preserve the safety, stability, and effectiveness of injectable medicines. Therefore, manufacturers treat the selection of the correct vial finish as an important step in pharmaceutical packaging design. The finish must remain compatible with the stopper, cap, or sealing mechanism used during filling and packaging processes. It must also allow healthcare professionals to access the medication safely while maintaining sterility.
Understanding the Serum Collar Finish for Pharmaceutical Vial Closures
Manufacturers widely use the serum collar finish in pharmaceutical packaging because it is specifically designed for injectable drugs. Serum collars work with rubber stoppers and aluminum seals to create a secure and airtight closure. Manufacturers produce these collars in standardized sizes such as 13 mm and 20 mm to ensure compatibility with various pharmaceutical filling and sealing machines. After filling the vial with medication, operators insert a rubber stopper into the opening and crimp an aluminum cap over it to hold the stopper firmly in place. This closure system protects the drug from contamination while allowing healthcare professionals to withdraw the medication by piercing the stopper with a syringe needle.
In addition to serum finishes, screw collar finishes feature threaded necks that allow users to seal vials with screw caps. This design makes them suitable for laboratory and research applications that require repeated access. Another option is the snap-on finish, which allows users to seal vials quickly and easily without specialized equipment and is commonly used for sample storage or non-sterile products. Regardless of the type, manufacturers must ensure that vial closure systems remain compatible with automated pharmaceutical manufacturing processes so that stoppering, sealing, and inspection operate efficiently. By selecting the appropriate closure and finish design, manufacturers ensure reliable packaging performance while maintaining the strict safety and regulatory standards required in the pharmaceutical industry.
Vials Based on Base Type
The base design of a vial plays a critical role in determining its stability, functionality, and suitability for different pharmaceutical and laboratory applications. The base structure directly influences how the vial performs during processes such as filling, processing, storage, and transportation. It also affects how securely the vial stands on surfaces and how smoothly it moves through automated pharmaceutical manufacturing equipment.
For this reason, pharmaceutical manufacturers carefully select vial base types according to the drug formulation, production requirements, and intended application. One of the most common designs is the concave or flat-bottom base, which is widely used for vials intended for liquid or powder filling. This design improves stability during manufacturing operations such as filling, stoppering, and sealing. The concave bottom allows the vial to sit firmly on conveyor systems and packaging equipment used in pharmaceutical production lines, reducing the risk of tipping and ensuring consistent performance during high-speed automated processes.
Types of Vial Bases Used in Pharmaceutical and Laboratory Applications
Manufacturers design another important base type specifically for lyophilization vials used in freeze-drying processes. Pharmaceutical companies commonly use lyophilization for sensitive products such as biologics, antibiotics, and vaccines. During this process, manufacturers freeze the drug solution and then dry it under low pressure to remove moisture while preserving the drug’s structure and stability. Because this process involves extreme temperature changes and vacuum conditions, manufacturers design lyophilization vials with specialized bases that withstand these stresses while maintaining structural integrity.
In addition to pharmaceutical applications, laboratories widely use base designs such as culture tubes and round-bottom tubes in diagnostic and research environments. These containers support efficient mixing, centrifugation, and sample handling during research and testing procedures. The base shape can also help liquids collect in a central area, allowing healthcare professionals to withdraw the full dose more easily with a syringe. Furthermore, standardized vial bases improve compatibility with automated inspection and packaging systems, enabling smooth movement through production lines while minimizing errors and improving overall manufacturing efficiency.
Components of Pharmaceutical Vials
Pharmaceutical vials used for injectable medicines are carefully designed to maintain sterility, safety, and product integrity throughout the drug’s lifecycle. These containers are not just simple bottles for storing medications; instead, they form part of a complete packaging system that protects sensitive drug formulations from contamination, leakage, and environmental exposure. Injectable vials typically consist of three essential components: the vial container, the rubber stopper, and the aluminum cap. Each component plays a specific role in maintaining the stability and sterility of the drug until it is administered. Proper compatibility between these components is critical to ensure container closure integrity and to meet strict pharmaceutical quality and regulatory standards.
Components of Pharmaceutical Vial Packaging

The vial container holds the medication, and manufacturers commonly make it from pharmaceutical-grade glass, such as borosilicate glass or from high-quality plastic materials that provide chemical resistance and structural stability. The rubber stopper, usually made from medical-grade elastomer materials like butyl rubber, acts as a protective barrier that prevents contamination and evaporation while allowing healthcare professionals to withdraw the medication with a syringe needle. Finally, the aluminum cap secures the stopper in place by crimping tightly around the neck of the vial, creating a strong mechanical seal that maintains sterility during storage and transportation. Together, these components form a reliable container closure system that protects injectable medicines, preserves drug stability, and ensures safe administration to patients.
Glass Vial Packaging
Glass vial packaging remains one of the most widely used and trusted solutions for storing injectable medicines in the pharmaceutical industry. These containers provide excellent chemical resistance and structural stability, making them highly suitable for sensitive drug formulations such as vaccines, biologics, and injectable antibiotics.
Pharmaceutical-grade glass, especially borosilicate glass, is chemically inert, which means it does not easily react with drug compounds. This property ensures that the container does not alter the composition, potency, or effectiveness of the medication stored inside. In addition, glass vial packaging creates a strong barrier against environmental factors such as oxygen, moisture, and contaminants, helping preserve drug quality and extend shelf life during storage and transportation.
Another important advantage of glass vials is their ability to withstand high-temperature sterilization processes commonly used in pharmaceutical manufacturing, including autoclaving and depyrogenation. These processes expose containers to extremely high temperatures to eliminate microorganisms, and glass materials can maintain their structural integrity under such conditions. Glass vials also offer high transparency, allowing healthcare professionals and quality control systems to visually inspect the contents for discoloration, particles, or other abnormalities before administration. Although glass containers can be fragile and require careful handling during transportation, their chemical stability, sterilization compatibility, and protective barrier properties make glass vial packaging one of the most reliable solutions for sterile pharmaceutical products.
Plastic Vial Packaging
Plastic vial packaging is becoming increasingly popular in pharmaceutical and laboratory applications, even though glass remains the dominant material for injectable drug containers. These containers offer several advantages, particularly where durability, lightweight design, and safe handling are essential. Unlike glass containers, plastic vials are highly resistant to breakage and are less likely to crack or shatter when dropped or exposed to mechanical stress.
This durability makes them safer for transportation, storage, and laboratory handling while reducing the risk of product loss caused by broken containers. In addition, plastic materials such as polypropylene provide good thermal insulation properties, helping maintain internal product temperature during short-term fluctuations. Their lightweight structure also reduces transportation and logistics costs, allowing pharmaceutical companies to ship large quantities of products more efficiently while making handling easier for healthcare professionals.
Advantages and Considerations of Plastic Vial Packaging
Modern manufacturing technologies enable manufacturers to produce plastic vials in a wide variety of shapes, sizes, and configurations to meet specific pharmaceutical requirements. Manufacturers can design containers with precise dimensions, integrated closures, and improved sealing systems that enhance usability and packaging efficiency. However, when selecting plastic containers for pharmaceutical products, manufacturers must carefully evaluate the compatibility between the plastic material and the drug formulation. Certain drug compounds may interact with plastic surfaces, which could lead to chemical reactions or absorption of active ingredients.
For this reason, manufacturers conduct extensive testing to ensure that plastic materials do not affect the stability or safety of the medication. Additionally, plastic containers may not tolerate high-temperature sterilization processes as effectively as glass containers, which limits their use in some sterile applications. Nevertheless, plastic vial packaging continues to gain popularity because of its durability, lightweight design, and cost efficiency, and advances in material technology are making plastic vials an increasingly viable solution for modern pharmaceutical packaging needs.
Applications of Pharmaceutical Vials
Healthcare professionals and laboratories widely use pharmaceutical vials across healthcare and laboratory environments. They commonly use these vials to store parenteral drugs, which healthcare professionals administer directly into the body without passing through the digestive system. Examples of parenteral drugs stored in vials include vaccines, insulin, antibiotics, and emergency medications. These drugs require containers that maintain sterility and prevent chemical interactions. Vials are also used extensively in laboratory diagnostics. Their precise dimensions and chemical resistance make them suitable for analytical instruments such as chromatography systems and autosamplers. In addition, pharmaceutical manufacturers use vials for nasal spray formulations and other specialized drug delivery systems.
Lyophilization and Pharmaceutical Vials
Manufacturers store many biologics and antibiotics in lyophilized form to improve their stability and shelf life. Lyophilization involves freeze-drying the drug under low pressure to remove water from the formulation.
During this process, the drug is first frozen and then dried through sublimation. Once freeze-dried, the medication can be stored for extended periods and reconstituted before administration.
However, the lyophilization process places significant stress on vial containers. Temperature changes and pressure variations can cause breakage or cosmetic defects. Therefore, pharmaceutical manufacturers use specially designed vials that withstand these conditions.
Pharmaceutical Vial Packaging Systems
Modern pharmaceutical facilities rely on highly automated equipment to maintain product quality and production efficiency. Pharmaceutical vial packaging processes include washing, sterilization, filling, sealing, inspection, and labelling.
These automated systems ensure consistent product quality and reduce the risk of contamination. Pharmaceutical vial packaging solutions are particularly important for high-volume production of vaccines and injectable medicines.
Vial Packaging Machinery and Automation

Modern pharmaceutical production relies heavily on advanced machinery to ensure accurate and efficient packaging processes. Vial packaging machinery performs multiple operations, including vial washing, sterilization, filling, stoppering, sealing, and inspection.
Vial filling machines are particularly important because they ensure precise dosing of sterile drug formulations. These machines operate in controlled environments to maintain sterility and prevent contamination.
In large-scale manufacturing facilities, manufacturers integrate multiple machines into complete vial packaging lines. These automated systems streamline production and maintain consistent quality across large batches of pharmaceutical products.
Complete Injectable Vial Liquid Packaging Line

The Complete Injectable Vial Liquid Packaging Line efficiently handles the packaging of liquid injectable products with high precision and reliability. The system handles 2 ml to 10 ml liquid vials. It integrates automated processes such as vial washing, sterilization, filling, stoppering, sealing, inspection, and labelling. These processes ensure smooth and continuous production. The line operates at speeds of up to 400 vials per minute. This speed helps pharmaceutical manufacturers achieve high productivity while maintaining strict sterility and quality standards. Advanced automation and precise control systems ensure accurate filling, consistent sealing, and reliable performance throughout the injectable vial packaging process.
Complete Injectable Dry Powder Vial Packaging Line

The Complete Injectable Dry Powder Vial Packaging Line efficiently processes and packages sterile dry powder formulations used in injectable pharmaceuticals. Designed for 2 ml to 100 ml vials, this advanced system integrates automated processes such as vial washing, sterilization, dry powder filling, stoppering, sealing, inspection, and labelling to ensure smooth and reliable production. Operating at speeds of up to 400 vials per minute, the packaging line helps pharmaceutical manufacturers achieve high productivity while maintaining strict sterility and quality standards. With precise dosing systems and advanced automation, the line ensures accurate powder filling, consistent sealing, and dependable performance throughout the entire dry powder vial.
Key Machines in a Vial Packaging Line
Individual machine categories within a complete vial packaging line include:
- Rotary or linear vial washing machines for container cleaning
- Sterilization tunnels for depyrogenation and drying
- Liquid or powder filling machines with precise volumetric/weight dosing
- Stoppering machines for rubber closure insertion
- Crimping / sealing machines for aluminium cap application
- Visual inspection systems
- Vial labelling machines for sticker application and coding
Regulatory Standards for Pharmaceutical Vials
Pharmaceutical vials and their packaging processes must comply with a comprehensive framework of international regulations and quality standards. Key requirements include:
- US FDA 21 CFR Parts 210 & 211 Current Good Manufacturing Practice (cGMP) regulations for finished pharmaceuticals, covering container-closure integrity, sterility assurance, and process validation.
- WHO Good Manufacturing Practices (WHO Technical Report Series). International GMP guidelines are widely adopted in emerging markets and referenced for prequalification programmes.
- USP <660> Containers Glass and USP <661> Plastic Packaging Systems United States Pharmacopoeia standards specifying chemical and physical requirements for pharmaceutical container materials.
- European Pharmacopoeia (Ph. Eur.) 3.2.1 Standards for glass containers for pharmaceutical use in European markets.
- ISO 15747 Standard for plastic containers for intravenous injections.
- ISO 11040 / ISO 11608 Standards covering prefilled syringes and needle-based injection systems that interact with vial packaging.
- ICH Q8/Q9/Q10 International Council for Harmonisation guidelines on pharmaceutical development, quality risk management, and pharmaceutical quality systems.
Compliance with these regulations ensures that vial containers, closures, and packaging processes consistently meet the highest quality and safety standards before the product reaches patients.
Conclusion
Pharmaceutical injectables play a critical role in modern healthcare. Because healthcare professionals administer them directly into the body, manufacturers must maintain strict safety and packaging standards. Vials help preserve the stability, sterility, and quality of injectable drugs.
Pharmaceutical vials come in a wide range of designs, materials, and sizes. They provide flexibility for storing liquid, powder, and lyophilized formulations. Combined with advanced packaging machinery and automated production systems, these containers help deliver life-saving medicines safely worldwide.
As pharmaceutical technology continues to evolve, improvements in vial design, materials, and packaging processes will further enhance drug safety, production efficiency, and patient outcomes.
Ultimately, vial packaging is a critical component of injectable drug delivery systems. It ensures that medicines remain sterile, stable, and protected throughout manufacturing, storage, transportation, and final administration. By combining reliable primary containers with protective secondary packaging solutions, pharmaceutical manufacturers can safeguard product quality while ensuring the safety of patients worldwide.

